Determining Phage Titer
Phage Titering is a procedure used to quantify the density of plaque-forming units (PFU, analogous to a bacterial cultures colony-forming units) in a given lysate. To achieve this, the researcher performing the experiment exposes exponentially growing bacteria to the bacteriophage of interest before allowing the bacteriophages to propagate overnight.Materials
- Phage lysate or freezer stock
- Top Agar (LB-Miller + 0.8% agar)
- LB-MIller (for diluting phage)
- LB-Agar plates (with antibiotic if appropriate)
- Small test tubes
- Heat block @ 60°C that can hold individual small test tubes
- Exponentially growing host cells
- Use exponential phase cells of a sensitive E. coli host strain prepared as described in the Phage Lysate Protocol.
Procedure
- Microwave flask of Top Agar to melt. Be sure to loosen the cap! For 250 ml of top agar ~15 min at power level 2 works well without any danger of boiling over.
- Allow agar to cool until handleable, then prepare 4 ml aliquots in small test tubes for as many samples as you will be handling. Place tubes in a heat block or water bath set at 60°C.
- Prepare dilutions of the phage stock in LB-Miller. Typically a dilution series from 100; to 1010 in 102 increments will give one plate with a countable number of plaques. These dilutions can be easily prepared by making 1.7 ml Eppendorf tubes with 990 µl LB and transferring 10 µl from the higher stock, mixing, and transferring to the next sample.
- Combine 100 µl of exponentially growing host cells and 100 µl of phage dilution in a fresh Eppendorf.
- Optional: Incubate unshaken at 37°C for 30 minutes.
- Add each sample to a top agar aliquot. Quickly but thoroughly mix by vortexing. You can vortex at full speed so the whirlpool reaches the bottom of the tube without it spilling out of the top. Pour the entire contents of test tube on top of an LB plate and rapidly rotate the plate around so that the agar covers the entire surface evenly before it cools.
- Let the plates cool ~15-30 minutes at room temperature and then transfer to an incubator at 30°C or 37°C.
Expected Results
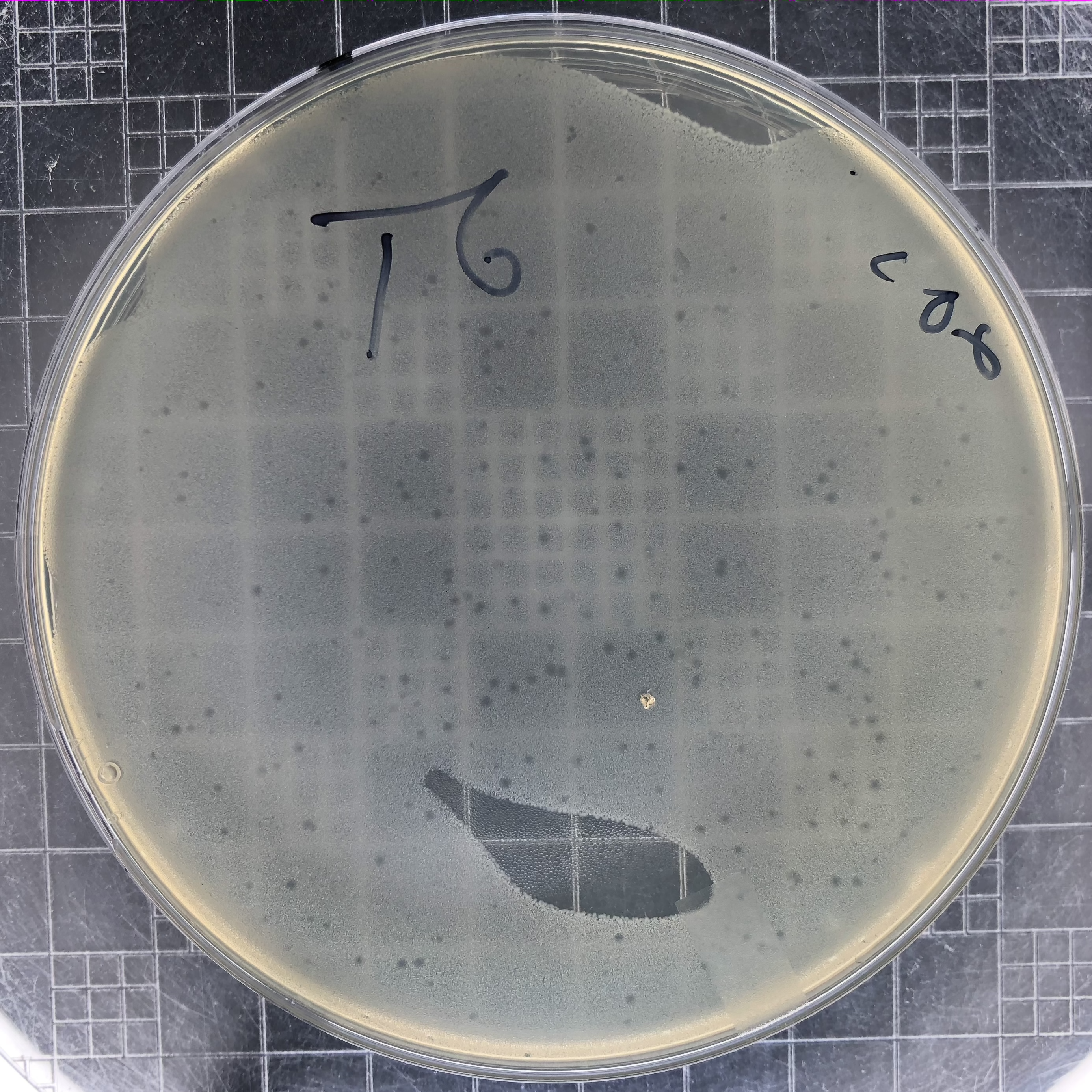 |
Phage T6 plaques after overnight growth at 37°C of a 106 dilution of lysate. Plaques for most E. coli T-even phages resemble these. They reach a maximum size and then stop spreading. Notice that the top agar was not spread to all places on top of this plate. |
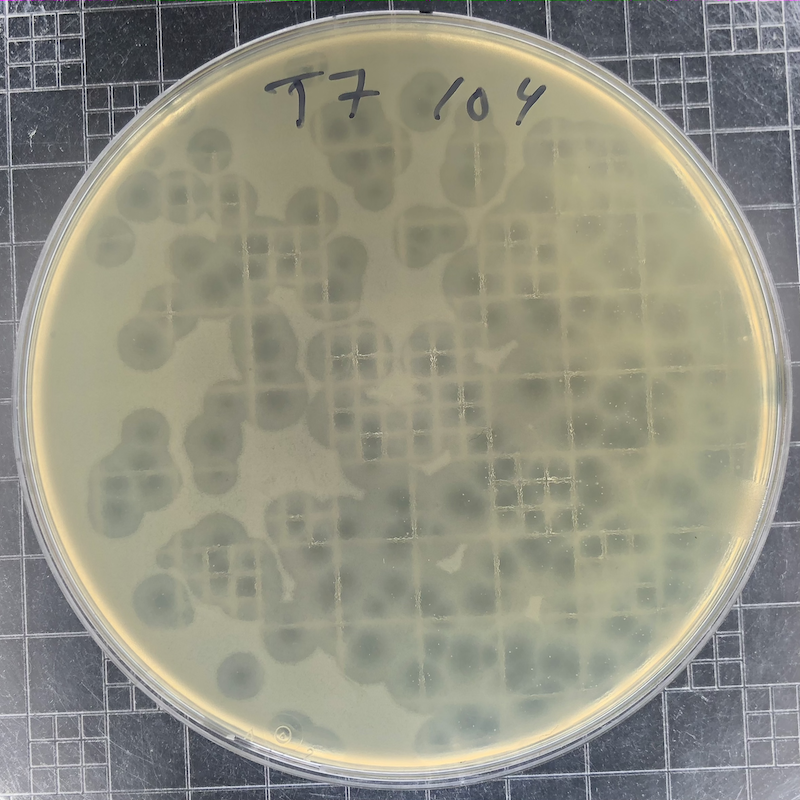 |
Phage T7 plaques after overnight growth at 37°C of a 104 dilution of a freezer stock. T7 plaques continue to grow. Incubate the plates at 30°C if you want to slow plaque growth. |
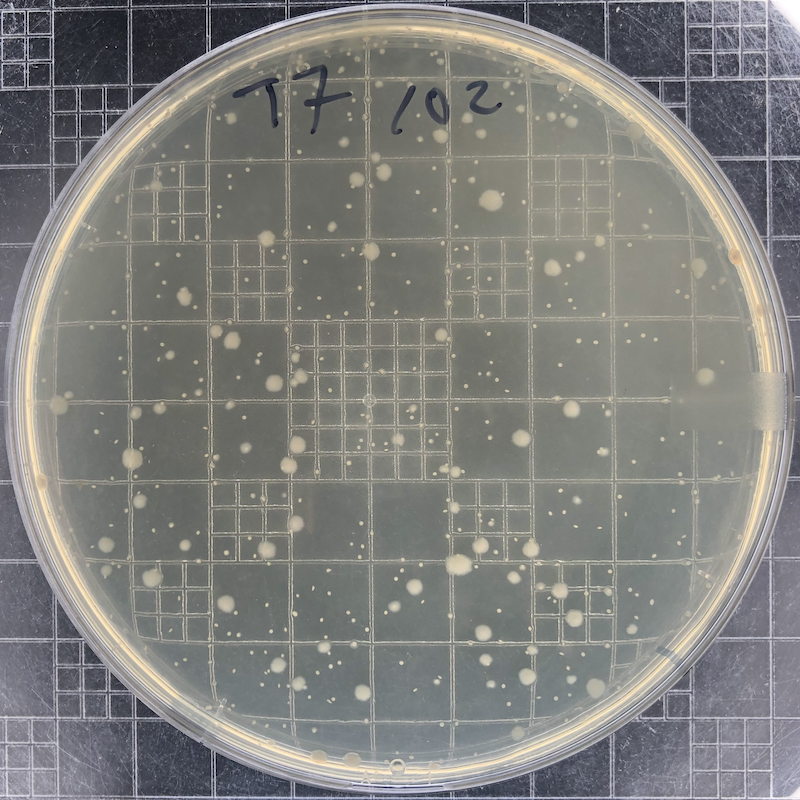 |
Phage T7 plaques after overnight growth at 37°C of a 102 dilution of a freezer stock. The phage has lysed all bacteria in this plate. The remaining colonies (small if they are embedded in the top agar and large if they reach the surface) are derived from mutant bacterial cells that have become resistant to the phage. |
Troubleshooting
- Top-agar layer is grainy or clumpy.
It likely cooled too much before you added phage+cells and poured it.
Example of Calculating Phage Titer
The final PFU count for each plate is n × 10 × d PFU/ml, where n is the number of plaques observed and d is the value from the dilution series. This is derived from the use of 0.1 ml of the phage stock to set up the initial dilutions. As an example, n=25 plaques on the d=106 dilution plate would represent 2.5×108 PFU/ml in the original lysis. Barrick Lab > ProtocolList > ProtocolsPhageAdsorptionRate > ProtocolPhageLysate > ProtocolsPhageTiters