| |
| META TOPICPARENT |
name="WebHome" |
<-- Preferences start here
- Set PAGETITLE = Barrick Lab :: Research
Preferences end here -->
Barrick Lab :: Research |
|
>
> | Listen to an introduction to our research: Studying Evolution and Engineering Bee Guts (EBRC in Translation Podcast) |
| |  See also: Previous Research Projects
See also: Previous Research Projects
Evolving and Engineering Insect Symbionts |
|
<
< | Many insects have more consequential associations with bacteria than we have with the human microbiome. We create genetic tools to study these bacterial symbionts and engineer their interactions with insects. We also use experimental evolution to examine how new symbionts arise and how interactions within a microbiome and between microbes and their hosts can change. These projects are highly collaborative and have also involved researchers from the Moran Lab, the Ellington Lab, the Davies Lab, and others. |
>
> | Many insects have more consequential associations with bacteria than we have with the human microbiome. We create genetic tools to study these bacterial symbionts and engineer their interactions with insects. We also use experimental evolution to examine how new symbionts arise and how interactions within a microbiome and between microbes and their hosts can change. These projects are highly collaborative and have also involved researchers from the Moran Lab, the Ellington Lab, the Davies Lab, and other research groups. |
| | |
|
<
< |  Current Funding: ARO MURI, NSF EDGE Current Funding: ARO MURI, NSF EDGE |
>
> |  Current Funding: ARO MURI, NSF EDGE, ERDC Past Funding: DARPA BRICS, DARPA Insect Allies Current Funding: ARO MURI, NSF EDGE, ERDC Past Funding: DARPA BRICS, DARPA Insect Allies |
| | |
|
<
< |  Past Funding: DARPA BRICS, DARPA Insect Allies Past Funding: DARPA BRICS, DARPA Insect Allies |
>
> |  Overview Publication Overview Publication |
|
>
> | |
| | |
|
<
< | Honey Bees: Protecting Pollinator Health |
>
> | Honey Bees: Functional Genomics and Protecting Pollinator Health |
| |
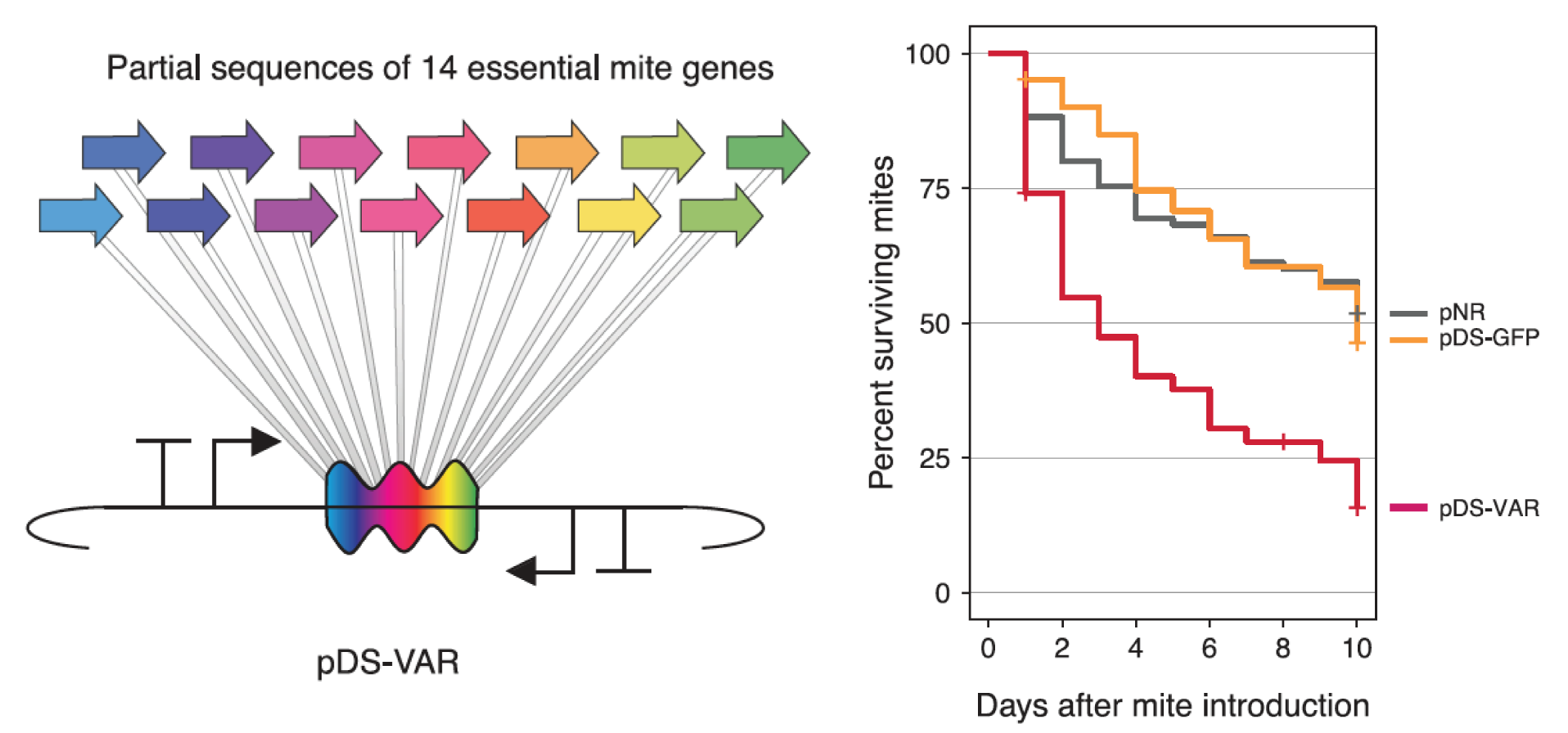
Engineered symbionts kill honey bee parasites
Bees are ecologically and economically important pollinators. We created a bee microbiome toolkit (BTK) for engineering bacteria that colonize the guts of honey bees ( Apis mellifera) and bumble bees ( Bombus species). We recently showed that we can engineer the bacterium Snodgrassella alvi to implement effective symbiont-mediated RNAi. This system can be used for the targeted knockdown of bee genes to study their functions. For example, we have used it to alter honey bee feeding behavior. We have also shown that these engineered symbionts can be used to protect honey bees from viral pathogens and arthropod parasites that imperil the health of hives. |
|
<
< |  Current Researchers: Zuberi Ashraf, Kadena Cope, PJ Lariviere, Dennis Mishler Current Researchers: Zuberi Ashraf, Kadena Cope, PJ Lariviere, Dennis Mishler |
>
> |  Current Researchers: Zuberi Ashraf, Kadena Cope, PJ Lariviere, Lucio Navarro, Dennis Mishler Current Researchers: Zuberi Ashraf, Kadena Cope, PJ Lariviere, Lucio Navarro, Dennis Mishler |
| | |
|
<
< |  Past Researchers: Sean Leonard Past Researchers: Sean Leonard |
| |  News: Bacteria Engineered to Protect Bees from Pests and Pathogens News: Bacteria Engineered to Protect Bees from Pests and Pathogens
 Representative Publications Representative Publications |
|
<
< | Aphids: Understanding the Evolution of an Endosymbiont |
>
> | Aphids: Understanding the Evolution of Endosymbionts |
| | |
|
<
< | 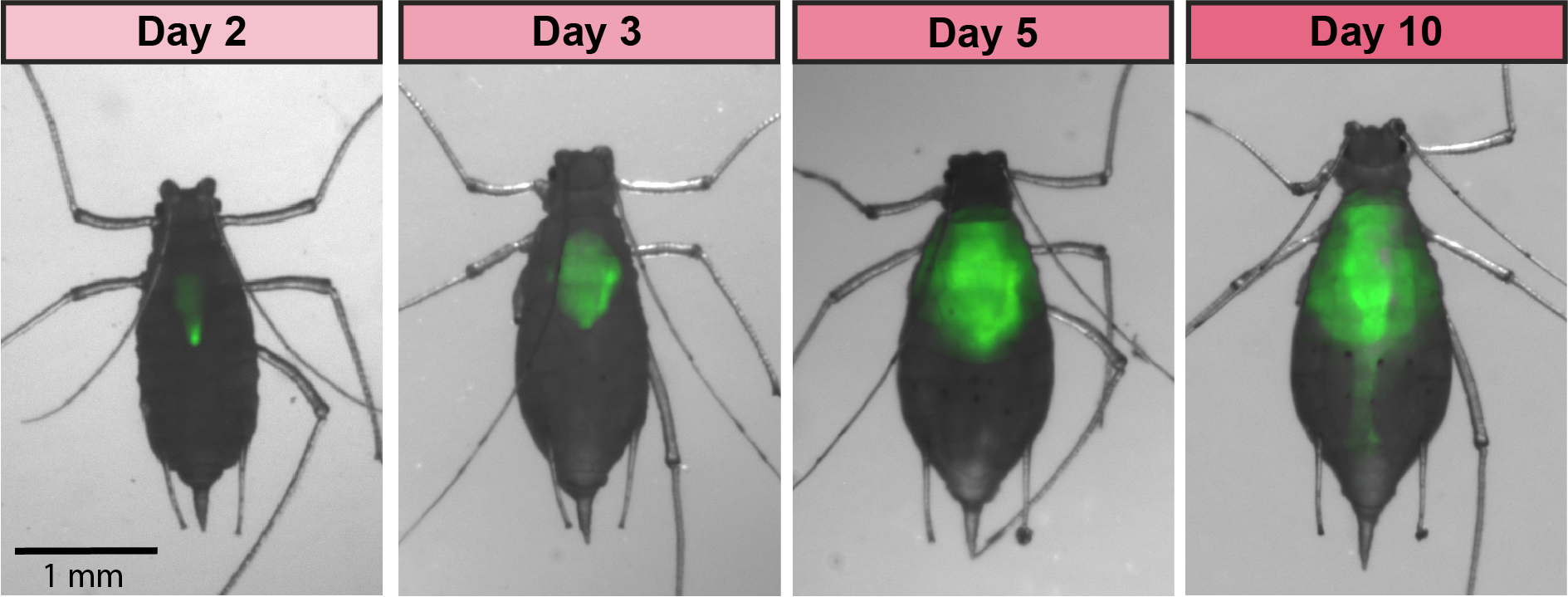
GFP-tagged symbiont colonizes the aphid gut |
>
> | 
GFP-tagged symbiont colonizing the aphid gut |
| |
Aphids are plant pests and model systems for studying bacteria-insect symbioses. We have developed tools for engineering strains of Serratia symbiotica that colonize the aphid gut. We are now using these tools to unravel how these strains, which can be pathogenic to their hosts, are related to strains of S. symbiotica that live within insect cells, are inherited across aphid generations, and can benefit their aphid hosts. We have showed that cultured S. symbiotica strains are capable of maternal transmission within aphids, suggesting that they possess a latent capacity to evolve a long-term symbiotic relationship with their hosts. We are also exploring applications of these engineered bacteria related to pest control.
 Current Researchers: Anthony VanDieren Current Researchers: Anthony VanDieren |
|
<
< |  Past Researchers: Kate Elston, Julie Perreau Past Researchers: Kate Elston, Julie Perreau |
| |
 News: Turning Plant Pests into Helpers News: Turning Plant Pests into Helpers
 Representative Publications Representative Publications
|
|
<
< | Leafhoppers: Studying and Engineering Natural Nanomaterials |
>
> | Leafhoppers: Evolution and Biochemistry of Natural Nanoparticles |
| |

Leafhopper and SEM image of brochosomes on its wing
|
|
<
< | Leafhoppers produce unique nanostructures, called brochosomes, that they anoint onto their wings and sometimes eggs. Brochosomes have interesting materials properties, including superhydrophobicity and omnidirectional antireflectivity. We are characterizing natural variation in brochosome structures and performing comparative genomics to understand the molecular components that make up these structures, how they evolved, and their biological functions. We are also working towards genetically engineering symbionts of leafhoppers to create new tools for studying brochosomes and potentially enhancing their properties for various applications. The Barrick lab leads an interdisciplinary grant that includes the Moran lab, Jewett lab, Schroeder lab, Alleyne lab, Milliron lab, and Freeman lab to study brochosomes. |
>
> | Leafhoppers produce unique nanostructures, called brochosomes, that they anoint onto their wings and sometimes eggs. Brochosomes have interesting materials properties, including superhydrophobicity and omnidirectional antireflectivity. We are characterizing natural variation in brochosome structures and performing comparative genomics to understand the molecular components that make up these structures, how they evolved, and their biological functions. We are also working towards genetically engineering symbionts of leafhoppers to create new tools for studying brochosomes and potentially enhancing their properties for various applications. |
| | |
|
<
< |  Researchers: Sarah Bialik, Kate Elston, Peng Geng, Elizabeth Robinson Researchers: Sarah Bialik, Kate Elston, Peng Geng, Elizabeth Robinson |
>
> |  Researchers: Jack Dwenger, Jace Gertz Researchers: Jack Dwenger, Jace Gertz |
| |
 News: Tiny Insects Provide Inspiration for New Biomaterials News: Tiny Insects Provide Inspiration for New Biomaterials |
|
<
< |
Preventing Evolutionary Failure in Synthetic Biology |
>
> |
Improving the Reliability of Synthetic Biology |
| |
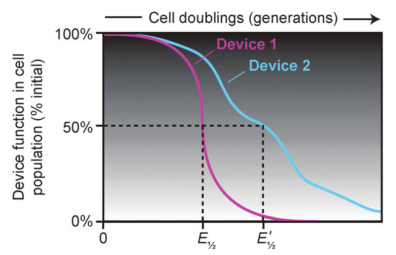
Evolutionary half-lives of biological devices
|
|
<
< | Synthetic biology applies engineering principles to create living systems with predictable and useful behaviors from collections of standardized genetic parts. However, living systems unlike mechanical devices inevitably evolve when their DNA sequences accumulate copying errors, often resulting in "broken" cells that no longer function as they were programmed. We are addressing this challenge by better characterizing how engineered cells evolve and using this information to design DNA sequences and host cells that are more robust against unwanted evolution. This work includes: (1) the development of the Evolutionary Failure Mode (EFM) Calculator software for identifying mutational hotspots in a designed DNA sequence; (2) using experimental evolution to identify "antimutator" variants of host organisms that lead to lower-than-natural mutation rates; and (3) designing genetic circuits that kill those cells within a population that are most likely to accumulate mutations. |
>
> | Synthetic biology applies engineering principles to create living systems with predictable and useful behaviors from collections of standardized genetic parts. However, living systems unlike mechanical devices inevitably evolve when their DNA sequences accumulate copying errors, often resulting in "broken" cells that no longer function as they were programmed. We are addressing this challenge by better characterizing how engineered cells evolve and using this information to design DNA sequences and host cells that are more robust against unwanted evolution. Also, precisely defining the function, extent, and provenance of genetic parts can be complicated because variation in these parts can arise from engineering or intentional evolution. This work includes: (1) developing "negative design" software to alert researchers to genetically unstable and unintentionally burdensome DNA designs; (2) using experimental evolution and engineering to create "antimutator" variants of host organisms that have lower-than-natural mutation rates; and (3) developing databases and software tools for improved annotation of engineered DNA sequences and common variants of those sequences. |
| | |
|
<
< |  Researchers: Matt McGuffie, Daniel Deatherage, UT Austin iGEM Team Researchers: Matt McGuffie, Daniel Deatherage, UT Austin iGEM Team |
>
> |  Current Researchers: Cameron Roots Current Researchers: Cameron Roots |
| | |
|
<
< |  Resources Resources |
| |  Representative Publications Representative Publications |
|
>
> | |
| | |
|
<
< |  Funding: NSF CBET, DARPA BRICS Funding: NSF CBET, DARPA BRICS |
>
> |  Current Funding: NIH R01; Past Funding: NSF CAREER, DARPA BRICS, DARPA Insect Allies Current Funding: NIH R01; Past Funding: NSF CAREER, DARPA BRICS, DARPA Insect Allies |
| |
Dynamics of Microbial Genome Evolution
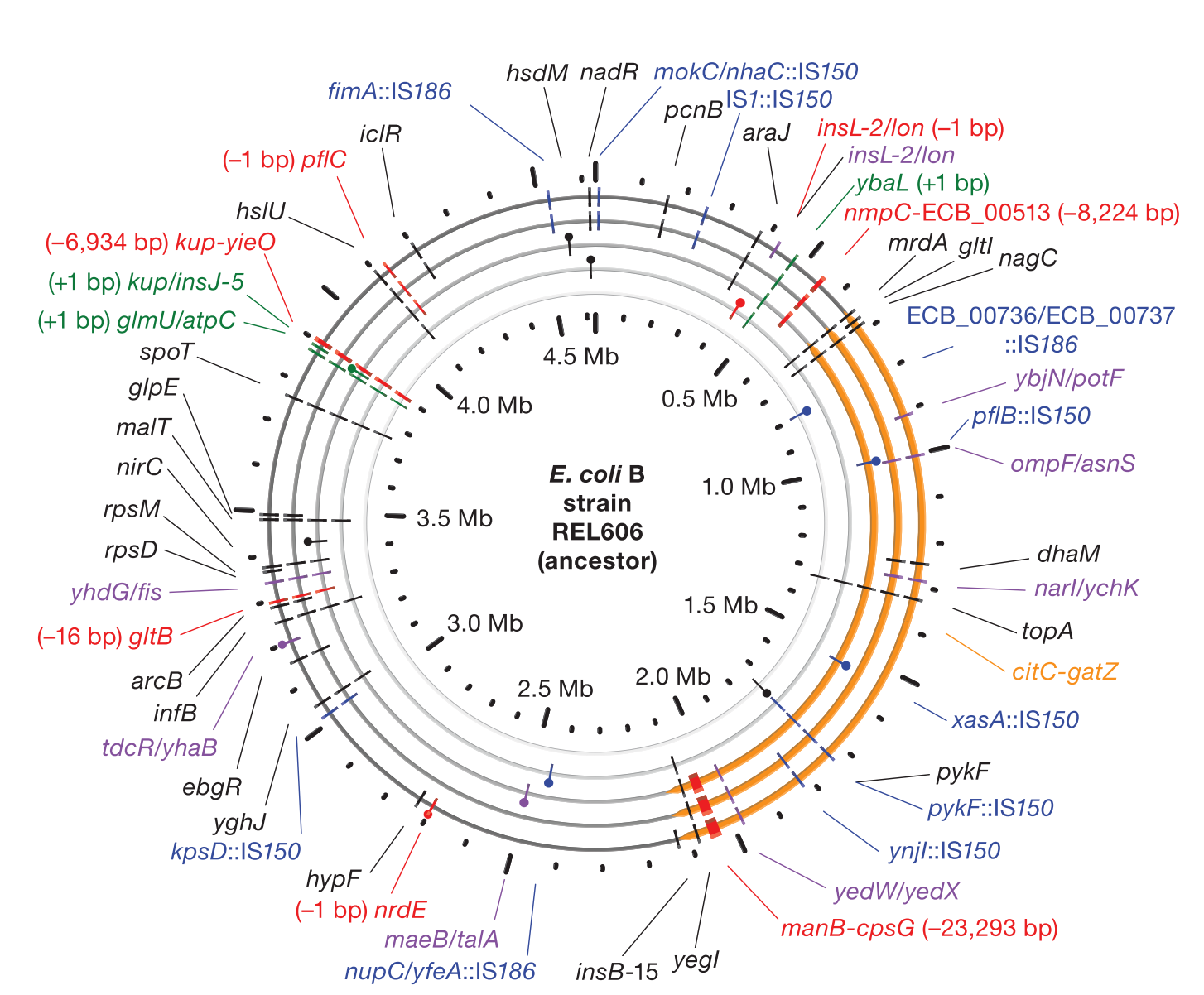
Accumulation of mutations in population Ara-1 of the LTEE over 20,000 generations of evolution
|
|
<
< | We develop the breseq computational pipeline for identifying mutations in laboratory-evolved microbial genomes from next-generation sequencing data. We have used this tool to extensively study rates of genome evolution in the 30-year Lenski long-term evolution experiment (LTEE) with E. coli. We continue to develop breseq so that it can be used for more additional applications related to strain engineering and medicine. For example, we are interested in how tracking rare variants within populations of microorganisms can anticipate further evolutionary trajectories and how this information might be used to better diagnose and treat disease. |
>
> | We develop the breseq computational pipeline for identifying mutations in laboratory-evolved microbial genomes from next-generation sequencing data. We have used this tool to extensively study rates of genome evolution in the 30-year Lenski long-term evolution experiment (LTEE) with E. coli. We continue to develop breseq so that it can be used for more additional applications related to strain engineering and medicine. For example, we are interested in how tracking rare variants within populations of microorganisms can anticipate further evolutionary trajectories and how this information might be used to better diagnose and treat disease. |
| | |
|
<
< |  Researchers: Daniel Deatherage Researchers: Daniel Deatherage |
>
> |  Current Researchers: Daniel Deatherage, Ira Zibbu, Jack Dwenger Current Researchers: Daniel Deatherage, Ira Zibbu, Jack Dwenger |
| | |
|
<
< |  Resources Resources |
>
> |  News: Legendary bacterial evolution experiment enters new era News: Legendary bacterial evolution experiment enters new era |
|
<
< | |
| |
 Representative Publications Representative Publications |
|
>
> | |
| | |
|
<
< | |
| |
- Deatherage and Barrick (2014) Methods Mol in Molecular Biology PMID:24838886
- Barrick and Lenski (2013) Nature Reviews Genetics. PMID:24166031
|
|
<
< |  Funding: NIH K99/R00, NSF, NSF BEACON Center, CPRIT Funding: NIH K99/R00, NSF, NSF BEACON Center, CPRIT |
>
> |  Funding: NSF LTREB, UT-CNS SPARK, NSF EEID, NASA Past Funding: NSF K99/R00 Funding: NSF LTREB, UT-CNS SPARK, NSF EEID, NASA Past Funding: NSF K99/R00 |
| | |
|
<
< | |
>
> | |
| | Evolution and Engineering of Naturally Transformable Bacteria
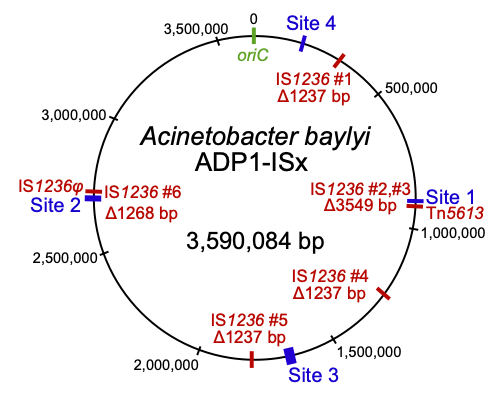
Insertion sequences (red) deleted in the transposon-free strain Acinetobacter baylyi ADP1-ISx
Naturally competent bacteria have expanded evolutionary potential because they can readily acquire new DNA from their environment. We are using experimental evolution of the model organism Acinetobacter baylyi ADP1 to understand how horizontally acquired genes and mobile genetic elements become domesticated after their incorporation into a new genome and the broader effects of gene acquisition on the rest of the genome. We are also studying the role of chemical specificity in determining the fate of acquired DNA as either nutrition or genetic information. These bacteria also provide an improved platform for studying microbial genome engineering due to the ease of reconstructing mutations and introducing new genes. We are investigating sources of genetic instability in ADP1 and engineering a clean genome version of this strain by deleting transposable elements and prophages. This will promote the use of ADP1 in synthetic biology by reducing rates of mutations that lead to inactivation of introduced genes. Further, we are using ADP1 as a platform to understand the limits to streamlining bacterial genomes and identifying adaptations to overcome the fitness costs of reduced genomes. |
|
<
< |  Researchers: Isaac Gifford Researchers: Isaac Gifford |
>
> |  Current Researchers: Isaac Gifford, Elizabeth Manriquez Current Researchers: Isaac Gifford, Elizabeth Manriquez |
| |
 Representative Publications Representative Publications |
|
<
< |  Funding: Welch Foundation Funding: Welch Foundation |
>
> |  Funding: NSF, UT-CNS SPARK Past Funding: Welch Foundation, NSF CAREER Funding: NSF, UT-CNS SPARK Past Funding: Welch Foundation, NSF CAREER |
| | |
|
<
< | |
>
> | |
| | Evolution and Engineering of Bacteriophages
Simulating engineered phage
The rise of multiple drug resistant pathogenic bacteria has raised the question of available, robust alternatives to antibiotics for disease treatment. Our lab is exploring new techniques for modifying bacteriophages for use in phage therapy. Evolution, engineering, and expansion of bacteriophage genomes has the potential to diversify target diseases for treatment, perform diagnostics for proactive disease prevention, and improve the efficacy of existing applications. We are working to accomplish these advancements through computational simulations and non-standard amino acid integration. The introduction of non-standard amino acids into the repertoire of protein production also brings insights into evolutionary outcomes that may not be possible in existing natural environments. Learning about evolution enabled by non-standard amino acids can lead to applications beyond the scope of viruses. We collaborate with the Wilke lab to use their Pinetree software for simulating phage gene expression and evolution. |
|
<
< |  Researchers: Peng Geng, Cameron Roots, UT Austin iGEM Team Researchers: Peng Geng, Cameron Roots, UT Austin iGEM Team |
>
> |  Current Researchers: Cameron Roots, Victor Li Current Researchers: Cameron Roots, Victor Li |
| |
 Representative Publications Representative Publications |
|
<
< | |
>
> | |
| | |
|
<
< |  Funding: NIH R01 Funding: NIH R01 |
>
> |  Funding: NIH R01 Past Funding: Welch Foundation Funding: NIH R01 Past Funding: Welch Foundation |
| |
| META FILEATTACHMENT |
attachment="collage.png" attr="h" comment="" date="1253739699" name="collage.png" path="collage.png" size="50520" stream="collage.png" tmpFilename="/usr/tmp/CGItemp40537" user="JeffreyBarrick" version="1" |
| META FILEATTACHMENT |
attachment="BacterialGrowthYield.pdf" attr="h" comment="" date="1263239270" name="BacterialGrowthYield.pdf" path="BacterialGrowthYield.pdf" size="74605" stream="BacterialGrowthYield.pdf" tmpFilename="/usr/tmp/CGItemp43365" user="JeffreyBarrick" version="1" |
| META FILEATTACHMENT |
attachment="DeepSequencingNucleicAcidSelection.pdf" attr="h" comment="" date="1263241294" name="DeepSequencingNucleicAcidSelection.pdf" path="DeepSequencingNucleicAcidSelection.pdf" size="79172" stream="DeepSequencingNucleicAcidSelection.pdf" tmpFilename="/usr/tmp/CGItemp43222" user="JeffreyBarrick" version="1" |
| META FILEATTACHMENT |
attachment="window_flasks.jpg" attr="h" comment="" date="1325974127" name="window_flasks.jpg" path="window_flasks.jpg" size="81708" stream="window_flasks.jpg" tmpFilename="/usr/tmp/CGItemp28487" user="JeffreyBarrick" version="1" |
| META FILEATTACHMENT |
attachment="eventual_winner_loser_fitness.png" attr="h" comment="" date="1377874213" name="eventual_winner_loser_fitness.png" path="eventual_winner_loser_fitness.png" size="28439" stream="eventual_winner_loser_fitness.png" tmpFilename="/usr/tmp/CGItemp44618" user="JeffreyBarrick" version="1" |
| META FILEATTACHMENT |
attachment="genome_circle_evolution.png" attr="h" comment="" date="1455894858" name="genome_circle_evolution.png" path="genome_circle_evolution.png" size="625247" stream="genome_circle_evolution.png" tmpFilename="/usr/tmp/CGItemp45984" user="JeffreyBarrick" version="1" |
| META FILEATTACHMENT |
attachment="citrate_metabolism.png" attr="h" comment="" date="1455894871" name="citrate_metabolism.png" path="citrate_metabolism.png" size="430811" stream="citrate_metabolism.png" tmpFilename="/usr/tmp/CGItemp45778" user="JeffreyBarrick" version="1" |
| META FILEATTACHMENT |
attachment="evolutionary_half_lives.png" attr="h" comment="" date="1455919624" name="evolutionary_half_lives.png" path="evolutionary_half_lives.png" size="84337" stream="evolutionary_half_lives.png" tmpFilename="/usr/tmp/CGItemp45719" user="JeffreyBarrick" version="1" |
| META FILEATTACHMENT |
attachment="Leonard_2018_ACS_Synth_Biol.gif" attr="h" comment="" date="1586439472" name="Leonard_2018_ACS_Synth_Biol.gif" path="Leonard_2018_ACS_Synth_Biol.gif" size="43362" stream="Leonard_2018_ACS_Synth_Biol.gif" tmpFilename="/usr/tmp/CGItemp49756" user="SarahBialik" version="1" |
| META FILEATTACHMENT |
attachment="Leonard_2020_Science.png" attr="h" comment="" date="1586439486" name="Leonard_2020_Science.png" path="Leonard_2020_Science.png" size="277053" stream="Leonard_2020_Science.png" tmpFilename="/usr/tmp/CGItemp49643" user="SarahBialik" version="1" |
| META FILEATTACHMENT |
attachment="PResERV.jpg" attr="h" comment="" date="1586443343" name="PResERV.jpg" path="PResERV.jpg" size="275915" stream="PResERV.jpg" tmpFilename="/usr/tmp/CGItemp57166" user="SarahBialik" version="1" |
| META FILEATTACHMENT |
attachment="Suarez_EnvMicrobiol_2017.png" attr="h" comment="" date="1586443996" name="Suarez_EnvMicrobiol_2017.png" path="Suarez_EnvMicrobiol_2017.png" size="345473" stream="Suarez_EnvMicrobiol_2017.png" tmpFilename="/usr/tmp/CGItemp57320" user="SarahBialik" version="1" |
| META FILEATTACHMENT |
attachment="website_fig_aphid.png" attr="h" comment="" date="1596740033" name="website_fig_aphid.png" path="website_fig_aphid.png" size="881458" stream="website_fig_aphid.png" tmpFilename="/usr/tmp/CGItemp49358" user="KateElston" version="1" |
| META FILEATTACHMENT |
attachment="leafhopper_website.png" attr="h" comment="" date="1596740897" name="leafhopper_website.png" path="leafhopper_website.png" size="489903" stream="leafhopper_website.png" tmpFilename="/usr/tmp/CGItemp49294" user="KateElston" version="1" |
| META FILEATTACHMENT |
attachment="ADP1_wiki_figure.png" attr="h" comment="" date="1596753530" name="ADP1_wiki_figure.png" path="ADP1_wiki_figure.png" size="64433" stream="ADP1_wiki_figure.png" tmpFilename="/usr/tmp/CGItemp40622" user="IsaacGifford" version="1" |
| META FILEATTACHMENT |
attachment="Pinetree_Phage_Glyph.png" attr="h" comment="" date="1597080445" name="Pinetree_Phage_Glyph.png" path="Pinetree_Phage_Glyph.png" size="115994" stream="Pinetree_Phage_Glyph.png" tmpFilename="/usr/tmp/CGItemp22733" user="CameronRoots" version="1" |
|









