Barrick Lab :: Research

- Active Projects
- Preventing Evolutionary Failure in Synthetic Biology
- Dynamics of Microbial Genome Evolution
- Identifying Mutations that Promote Bacterial Evolvability
- Evolving and Engineering Naturally Transformable Bacteria
- Experimental Evolution with Expanded Genetic Codes
- AG3C: Associating Growth Conditions with Cellular Composition
- Previous Projects
- Other Interests
Active Projects
Preventing Evolutionary Failure in Synthetic Biology
*In progress* Resources Representative Publications- Jack et al. (2015) ACS Synthetic Biol. PMID:26096262
- Renda et al. (2014) Mol. Biosyst. PMID:24556867
Dynamics of Microbial Genome Evolution
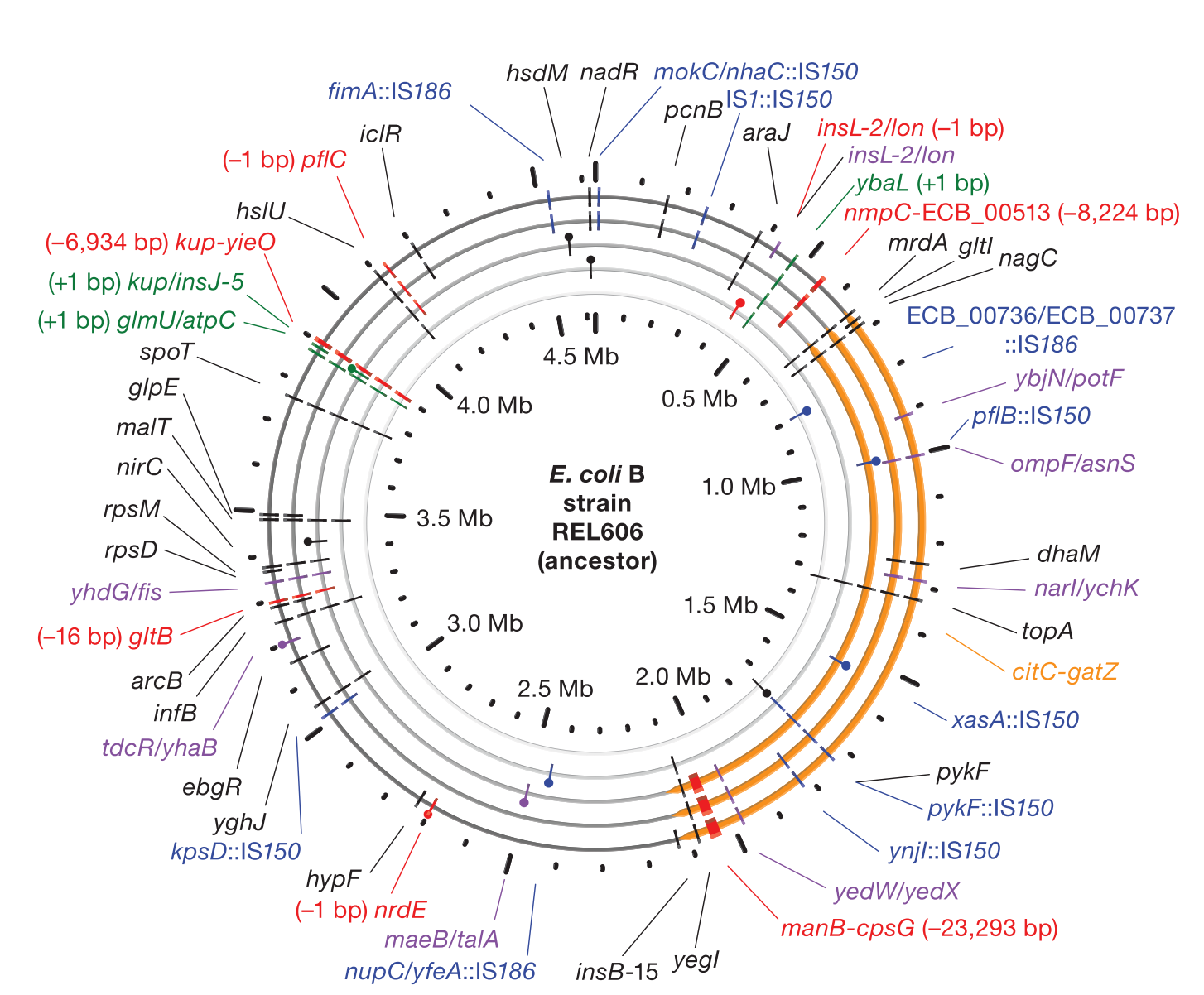
- Tenaillon et al. (2016) Preprint. bioRxiv:036806
- Deatherage and Barrick (2014) Methods Mol. Biol PMID:24838886
- Barrick and Lenski (2014) Nat. Rev. Genet. PMID:24166031
Identifying Mutations that Promote Bacterial Evolvability
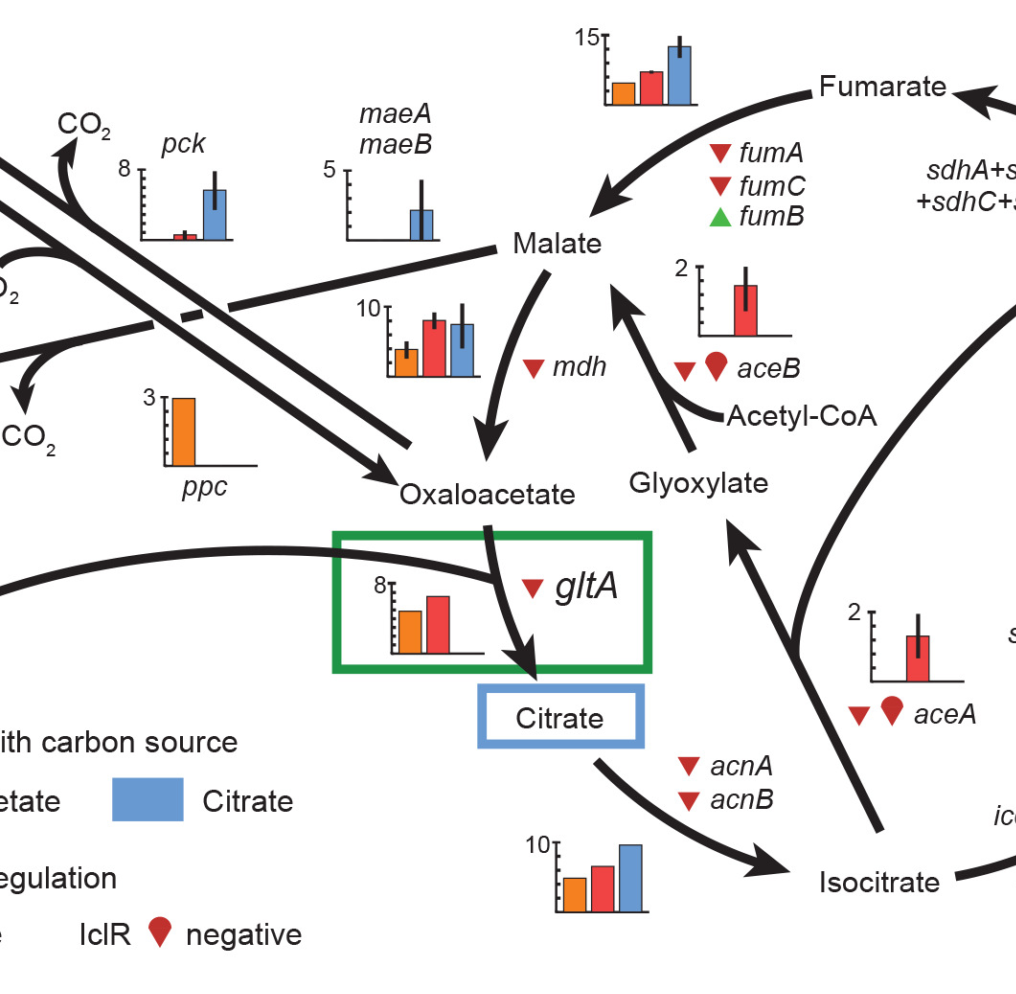
Evolutionary potential (or evolvability) is the capacity of an organism to generate descendants with increased fitness or new adaptive characteristics. Evolvability is a complex trait. It depends not only on how developmental and regulatory processes render underlying genetic changes into phenotypic variation, but also on the dynamics of how mutations arise and compete within a population. Mutator strains of microorganisms, which have elevated genomic mutation rates due to defects in DNA proofreading or repair pathways, can sometimes take over pathogen populations because they promote evolvability. However, little is currently known about how mutations affecting other cellular processes impact microbial evolvability or about how evolvability varies at each step in a typical adaptive trajectory. We are currently reconstructing the dynamics of mutations in populations from a Lenski 25-year long-term evolution experiment (LTEE) with _E. coli_ to understand two cases of evolvability differences not related to mutation rates: In the first, recurring mutations in a specific gene that were very beneficial in the short term restricted further evolvability to the extent that these lineages always went extinct later. 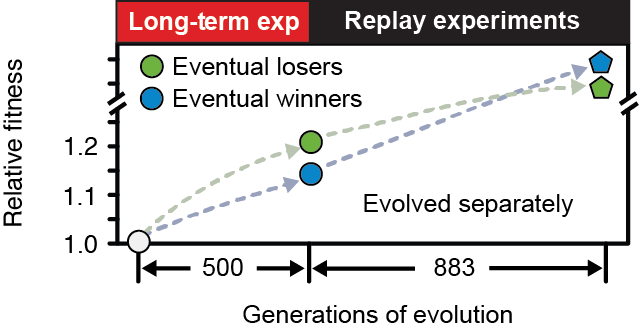 E. coli Eventual Winners and Losers. Bacteria that had evolved higher fitness early in a population reproducibly lost later when evolution was replayed many times from each starting point to competitors with different, less-beneficial mutations. The eventual losers apparently had favorable mutations that reduced further evolvability. |
 Epistasis Poker Analogy. Early draws of cards that are more beneficial to the value of a poker hand (analogous to the most beneficial mutations in the eventual loser E. coli), prevent them from reaching the best hand after more cards are drawn. |
- Quandt et al (2015) eLife PMID: 26465114
- Quandt et al (2014) PNAS PMID:24379390
- Blount et al (2012) Nature PMID:22992527
- Woods et al. (2011) Science PMID:21415350
Evolving and Engineering Naturally Transformable Bacteria
Naturally competent bacteria may have increased evolutionary potential because they can directly acquire new genes from their environments and incorporate them into their genomes. In addition to this possibility of a new mutational move in genotype-phenotype space (by horizontal gene transfer), these microbes provide an improved platform for studying microbial genome engineering and evolution due to the ease of reconstructing mutations and introducing new genes. We are investigating sources of genetic instability in the naturally competent bacterium Acinetobacter baylyi ADP1 and engineering a clean genome version of this strain to have reduced rates of mutations that lead to inactivation of introduced genes for synthetic biology applications. We are also using experimental evolution to test for the utility of providing foreign DNA sequences and to understand how horizontally acquired genes become domesticated after they are incorporated into a new genome. Finally, we are using ADP1 as a platform for understanding the limits of simplifying a bacterial genome by evolutionary streamlining approaches. Representative Publication- Renda et al. (2015) J. Bacteriol. PMID:25512307
Experimental Evolution with Expanded Genetic Codes
Another way to potentially increase evolvability is to augment the chemical diversity of the building blocks available to an organism. We are evolving microorganisms, including bacteriophage, in the context of expanded genetic codes that re-code the amber stop codon into various non-canonical amino acids. This change increases the sequence-structure-function space available for evolution to explore. We are studying how evolution must compensate for the global change in how the information in an organism's genome is decoded, in cases where it is possible for an organism to survive this transition, and also how dependence on the new amino acid develops over time, such that an organism can no longer survive in the context of a normal genetic code. Finally, we are systematically examining how the unique chemistries of different amino acid building blocks are tolerated and uniquely useful for adaptation and using this technology to recapitulate the steps in a hypothesized pathway in which new post-translational modifications evolve in a series of adaptive steps from nonenzymatic metabolite damage to the proteome. Representative Publication- Hammerling et al. (2014) Nat. Chem. Biol. PMID:24487692
AG3C: Associating Growth Conditions with Cellular Composition
We are part of a collaboration of eight labs (including experimental biologists, computational biologists, and mathematicians) that is collecting and analyzing comprehensive transcriptomics, proteomics, lipidomics, and metabolic flux data from bacterial samples grown under different conditions. The aim is to solve the "inverse problem" of predicting the conditions that were used to culture a microbial sample from measurements in these high-dimensional data sets (AG3C Website). Our lab is primarily responsible for creating the microbial samples and using RNA-seq to examine RNA expression levels. Representative Publication- Houser et al. (2015) PLoS Comput. Biol. PMID:26275208
Previous Projects
Discovering Functional Nucleic Acid Families by Deep Sequencing and Fold Sampling
A final way that we are studying how to improve the potential of evolutionary methods is in systems for the in vitro selection of nucleic acid molecules. Nucleic acids with random sequences often misfold into inactive structures that are kinetically trapped. That is, they become stuck and cannot unfold to rearrange into other, potentially active, structures on a reasonable timescale. It is possible that this potential for misfolding, which is known to occur for even natural ribozymes, limits the recovery of new families of functional nucleic acids (aptamers, ribozymes, deoxyribozymes, riboswitches) by laboratory evolution methods that select for functional molecules from pools of 10131015 molecules with randomized sequences. We expect this problem to more greatly affect longer nucleic acid sequences, which have the capacity for folding into elaborate three-dimensional structures that achieve more rapid rates of catalysis or tighter binding affinities. We are currently using next-generation deep-sequencing techniques and methods to allow a single nucleic acid strand to have multiple chances to fold correctly to pass selection to see if they can enhance our ability to recover more complex families of functional nucleic acids. Representative Publications- Reba et al. (2012) Artificial Life XIII PMID:25432719
Other Interests
Evolution Experiments with Digital Organisms
Populations of self-replicating computer programs mutate and compete for CPU cycles in the Avida artificial life system. This environment for digital organisms has been used to ask fundamental questions that transcend the substrate that is evolving. The speed of digital evolution experiments, transparency of the underlying mechanics, and ability to manipulate and record every aspect of the evolutionary process make it possible to more readily explore cause and effect relationships in Avida than in natural systems. I have extensive experience with Avida. My lab will use Avida to complement studies in biological systems: both as a way of more clearly illustrating and exhaustively characterizing phenomena of interest and as a source of new candidate laws and insights to motivate new lines of experimental inquiry. ResourcesCollaboration Interests
In addition to these specific plans for future research, we have long-standing interests in RNA biochemistry, riboswitches and other noncoding RNAs in bacteria, and comparative genomics. We hope to be a resource to colleagues who are interested in adding an evolutionary, informatics, or genomics dimension to their studies. Whenever possible, we would like to investigate our basic research questions about evolution in systems where there is additional interest in the underlying molecules, cells, and mutations. We are eager to link up with researchers and those in industry who have relevant problems in medicine and biotechnology to find these opportunities. Please contact Prof. Barrick about potential collaborations. Barrick Lab > WebLeftBar > ResearchInterests
Topic revision: r27 - 2016-02-19 - 21:19:52 - Main.JeffreyBarrick